ABOUT DEUTERIUM
A TERSE TUTORIAL.
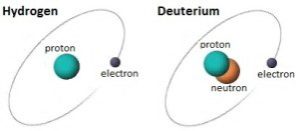
Hydrogen is the most common element on earth. It’s far more plentiful than oxygen, carbon, calcium and all of those other geeky elements we learned about in school. It’s also absolutely essential for life and is in the air we breathe, the food we eat and the water we drink. The hydrogens that we take into our body become part of blood, organs, DNA and everything else that makes us us. Deuterium is a rare type of hydrogen – only 155 out of every million hydrogens are deuterium. It contains a proton and a neutron in its nucleus. The extra neutron doubles the mass of this type of hydrogen atom. The symbols for deuterium are 2H and D.
When two deuteriums combine with oxygen, the resulting water is referred to as “heavy” water 2H2O because of the additional mass. It’s called semi-heavy water,2HOH, when one normal atom of hydrogen and one atom of deuterium combine with oxygen. Water made with deuterium tastes and looks the same as normal water but has numerous distinguishing characteristics.
For example:
- Normal water boils at 100° C; heavy water boils at 101.4 °C.
- Normal water freezes at 0° C; heavy water freezes at 3.8° C.
- Ice normally floats on the surface of water; ice made with heavy water sinks.
The effects that deuterium’s extra mass has on water demonstrates that it can make it react differently from normal hydrogen in chemical reactions. Deuterium can also has the same type of effects on your metabolism.
Here are several examples:
- It decreases your energy production.
- It decreases your ability to use energy.
- It deforms the three dimensional structure of your proteins, fats and carbohydrates you eat and your body makes.
- It doesn’t allow your DNA to properly fold and replicate.
- It changes the binding constants of your enzymatic reactions.
- It changes the pharmacokinetics and pharmacodynamics of the medicines, supplements and other therapeutics you take.
- It may ultimately control who you are and how you respond to clinical treatments.
These kind of isotopic effects can have dire metabolic consequences!
HOW DEUTERIUM DESTROYS YOUR METABOLISM
WHAT DEUTERIUM MEANS TO YOUR HEALTH AND PERFORMANCE.
Think of your body as a reservoir. It simply holds what you give it and uses it as you need it.
Now, picture your body as having two sources of water – the water that you drink and the water that your mitochondria make from the food you eat. Different water sources have varying levels of deuterium content from its source. The water you drink that is in sodas, etc. have different, typically much higher, deuterium content as well.
As a consequence, the deuterium content in your blood and tissues is from the combination of the deuterium in the water you drink and the water made by your mitochondria from what you eat.
Now why is your deuterium level so important to your health?
The most important isotope we encounter through food and liquids is deuterium, the heavy isotope of hydrogen atoms, and hydrogen ions drive and spin our energy producing nanomotor, ATP synthase, by an incredible ~1500 hydrogen ions transferred per second. See how it works.
As heavy deuterium isotopes break this delicate protein in our cells, it may cause metabolite crowding, which sets the stage for population diseases, which may affect you.
Our battery of diagnostic tests allows us to see what deuterium level you have in your body and what kind of havoc it is reaping on your metabolism. We can then suggest a medical food protocol, supplement and/or deuterium depleting water that will bring it down to a maximum target range, which is the level that the body has mechanisms in place to effectively control and deplete.
For general health and disease risk evaluations, take advantage of the 2Health Clinic’s deuterium depletion and metabolic restoration efforts to improve your health.
If your body can’t deplete deuterium on its own, we need to look deeper, and that’s the 2Health Clinic’s story.
And this is the story about your nanomotors.

